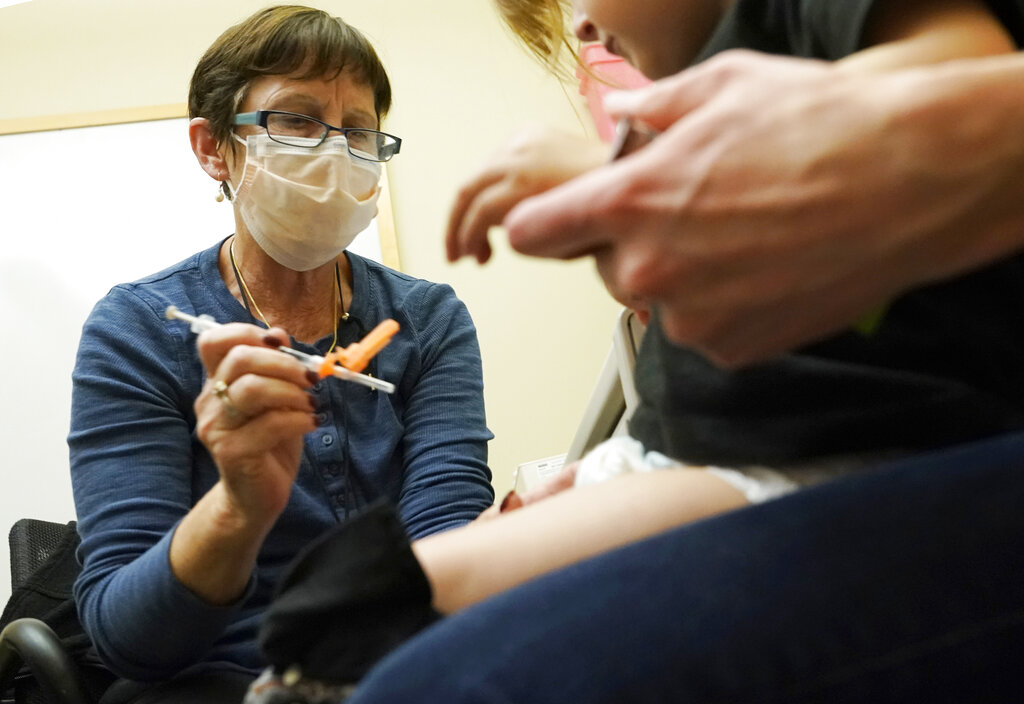
A brand new way of treating leukemia is on its way to the U.S. market, and it could change how doctors fight cancer.
The Food and Drug Administration approved a treatment called Kymriah. It's designed to fight a type of acute lymphoblastic leukemia, the most common childhood cancer.
It works by taking cells out of the patient's body and modifying them to fight cancer cells, then reintroducing them to the immune system in a process known as gene therapy. The treatment is the first of its kind approved in the U.S.
There's plenty of reason to be excited about Kymriah. The FDA says it provides a sorely needed treatment option for the disease.
It also appears to be effective. A clinical trial found 83 percent of patients who underwent the treatment went into remission within three months.
But there are drawbacks — the main one being the price tag. The treatment costs $475,000.
Despite the high price tag, it's worth remembering that other leukemia treatments, like bone marrow transplants, can be even more expensive.
The drug's maker, Novartis, also pledged that it wouldn't charge any patient who didn't respond to the treatment after a month.
The treatment also has some potential side effects, including an immune system overreaction known as cytokine release syndrome and low blood pressure.
After the FDA approval, hospitals are close to offering Kymriah to patients. Dr. Kevin Curran, an oncologist who hopes to use the treatment soon, told The New York Times: "This is a big paradigm shift, using this living drug. It will provide a lot of hope. This is the beginning."




 Where You Live Could Affect Your Cancer Risk
Where You Live Could Affect Your Cancer Risk